Imagine taking a medication that works perfectly on an empty stomach but becomes almost useless-or dangerously potent-after a breakfast burrito. This isn't a hypothetical scenario; it's a primary concern in pharmaceutical development. Whether you are a researcher, a regulator, or someone interested in how the body processes chemicals, understanding the difference between fasted and fed states is the key to ensuring a drug is both safe and effective for everyone, regardless of when they last ate.
At its core, bioequivalence standards is the process of ensuring that a generic drug performs the same way in the body as the brand-name version. To do this accurately, scientists can't just test a drug once. They have to look at two distinct environments: the fasted state and the fed state. If a drug's absorption changes drastically based on the presence of food, it can lead to dosing errors or unexpected side effects.
| Attribute | Fasted State | Fed State |
|---|---|---|
| Gastric Residence Time | Average 13.7 minutes | Average 78.3 minutes |
| Intragastric pH | Median minimum 2.5 | Median minimum 1.5 |
| Free Fatty Acid (FFA) Availability | 30-50% Higher | Lower |
| Gastric Pressure | 30-304 mbar (variable) | Consistently > 240 mbar |
The Mechanics of the Fasted State
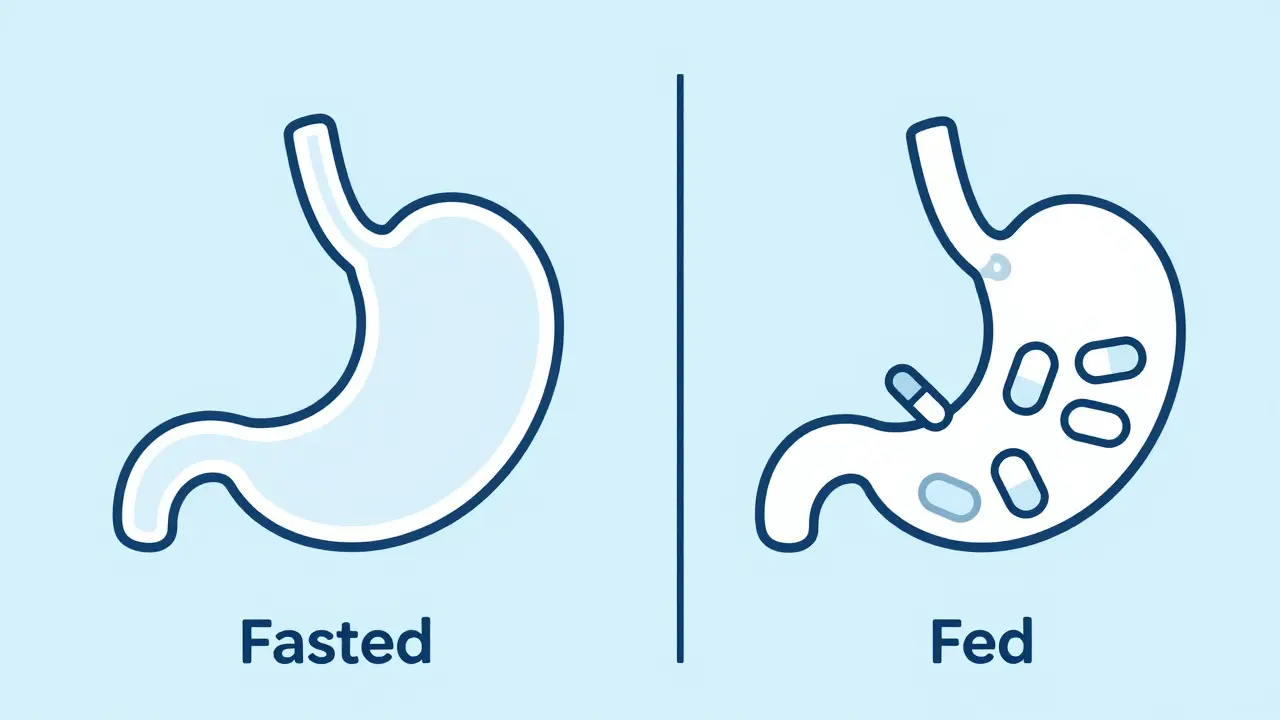
In clinical trials, a Fasted State is a physiological condition where a subject has abstained from caloric intake for typically 8 to 12 hours . During this window, the body shifts its metabolic focus. Since there is no incoming glucose from a meal, the body ramps up lipolysis, increasing the availability of free fatty acids to use as energy. For a drug, this means the stomach is essentially an empty tunnel. The drug moves quickly from the stomach into the small intestine, where most absorption happens.
Testing in this state provides a baseline. It tells researchers the maximum speed of absorption and the drug's natural behavior without interference. However, relying solely on fasted data is risky. If a drug is highly lipophilic-meaning it dissolves easily in fats-it might be absorbed poorly on an empty stomach but absorbed rapidly when fats are present, potentially leading to a "spike" in blood concentration that could be toxic.
Why the Fed State Changes Everything
The Fed State is the postprandial period, usually 2 to 4 hours after consuming a standardized meal . When you eat, your gastrointestinal tract transforms. The stomach doesn't just hold food; it changes its pH and slows down the rate at which it empties its contents into the intestine. This is known as gastric residence time. According to SmartPill capsule studies, the time a substance spends in the stomach jumps from about 14 minutes (fasted) to nearly 78 minutes (fed).
This delay can be a double-edged sword. For some drugs, a slower release is beneficial. For others, the increased acidity (pH dropping to 1.5) can degrade the medication before it even reaches the absorption site. Furthermore, the FDA (U.S. Food and Drug Administration) defines a specific "high-fat, high-calorie meal" for these tests-usually around 800 to 1,000 calories with about 500-600 calories coming from fat. This rigorous standard ensures that the "food effect" is tested under the most challenging conditions possible.
The Impact on Drug Bioavailability
The real-world goal of these tests is to measure Bioavailability, which is the proportion of a drug that enters the circulation and is able to have an active effect. Food can swing this number wildly. For example, lipophilic compounds like fenofibrate can see a 200-300% increase in bioavailability when taken with food. On the flip side, drugs like griseofulvin can see absorption drop by 50-70% in a fed state.
This is why the EMA (European Medicines Agency) requires fed-state testing for almost all oral drugs if the food effect is unknown. Their data shows that roughly 35% of drugs show clinically significant interactions with food. If a doctor tells a patient to "take this on an empty stomach," that instruction is based directly on the results of these dual-condition tests.
Beyond Pharmaceuticals: The Exercise Perspective
While the stakes in medicine are about safety and dosing, the same fasted vs. fed dichotomy plays out in sports science. The difference here is performance versus adaptation. A meta-analysis of 46 studies showed that exercising in a fed state can improve prolonged aerobic performance by about 8.3%. This is because the body has a ready supply of glycogen, allowing athletes to sustain higher intensities for longer.
However, training in a fasted state triggers different biological switches. It upregulates PGC-1α, a protein that helps create new mitochondria in the cells. This leads to better fat-burning efficiency and improved insulin sensitivity. Many endurance athletes use a "periodized" approach-training fasted on some days to improve metabolic health and training fed on others to hit peak power outputs. It's a balance between burning fat and performing at the limit.

Pitfalls and Practical Implementation
Simulating a "real-world" meal is harder than it sounds. People don't all eat the same things, and their bodies don't all react the same way. Recent research highlights that ethnicity plays a role; for instance, some studies indicate that Asian subjects may have gastric emptying times that are 18-22% slower than Caucasian subjects in fed conditions. This is why the FDA is now expanding requirements to include more diverse populations in their trials.
For a study to be valid, researchers must control for variables that could skew the data. This includes:
- Sleep duration: Ensuring subjects have at least 7 hours of sleep to avoid metabolic stress.
- Hydration: Checking urine specific gravity (usually < 1.020) to ensure the subject isn't dehydrated.
- Standardization: Keeping the high-fat meal within ±10% of the specified caloric and macronutrient values.
The Bottom Line on Dual Testing
Skipping either the fasted or fed state in a study is like trying to understand a coin by only looking at one side. You miss the full picture of how a substance interacts with the human body. Whether it's ensuring a generic drug is bioequivalent to a brand-name one or optimizing a marathon runner's diet, the interaction between nutrients and chemistry is too powerful to ignore.
What is the standard definition of a fasted state for clinical testing?
In most pharmaceutical and physiological studies, a fasted state is defined as having no caloric intake for a period of 8 to 12 hours. During this time, only water is typically permitted to ensure that the gastrointestinal tract is clear and the body's insulin levels are low.
How does food specifically affect drug absorption?
Food affects absorption in three main ways: it can change the pH of the stomach, slow down the speed at which the drug reaches the small intestine (gastric emptying), and provide fats that help lipophilic drugs dissolve and enter the bloodstream more easily.
Why do the FDA and EMA require both fasted and fed studies?
They require both to prevent dosing errors. Because food can either either increase bioavailability by 300% or decrease it by 70%, a drug that is safe when fasted might become toxic or ineffective when taken with a meal. Dual testing allows for precise labeling instructions.
Is fasted exercise always better for fat loss?
Not necessarily. While fasted exercise increases the immediate oxidation of fats and boosts certain mitochondrial proteins, long-term studies on body composition often show little difference compared to fed-state training over several weeks. The primary benefit is metabolic adaptation, not necessarily faster weight loss.
What is a "high-fat meal" in the context of bioequivalence?
According to FDA guidelines, a high-fat meal typically consists of approximately 800-1,000 calories, where about 50-60% of those calories (roughly 500-600 calories) come from fat. This is used to simulate a "worst-case" scenario for food-drug interactions.

Written by Felix Greendale
View all posts by: Felix Greendale